Progress in ionic local structures of high-temperature FLiBe molten salts
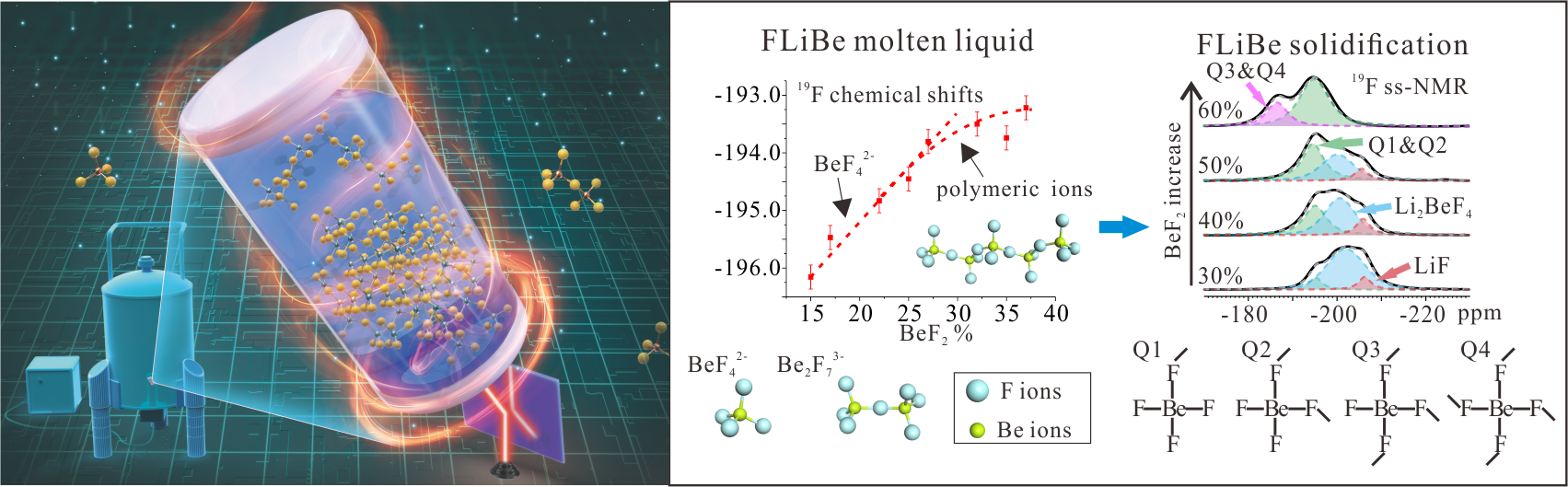
As an effective heat transfer fluid at high temperature, low pressure, and high neutron radiation flux, the LiF-BeF2 (FLiBe) salts are selected as primary coolant or fuel carrier in Thorium Molten Salt Reactor. In the molten salts, many multiply charged metal ions forming coordinated local structures have important effects on their physical and chemical properties, such as liquid alkali halide, and can adjust the viscosity, density, and other salt thermophysical properties by their diffusion and motion. Due to the toxicity, volatilization of beryllium fluorides, the in-situ experiments on the molecular structure of FLiBe are rarely reported without high-temperature and high-resolution analytical methods.
In this work, the local structure of FLiBe melts was conducted by the high-temperature nuclear magnetic resonance spectroscopy (HT-NMR), the strong covalent interactions of Be2+ ions can facilitate the coordination between Be2+ ions and F- ions, and the polymeric Be-F chains and networks are closely related to the ionic dynamics. Also, the solid FLiBe eutectic salts at ambient temperature are analyzed by 1D and 2D solid-state NMR (ss-NMR), thus verifying that the network structures of FLiBe are similar as those of silicate glasses. The NMR method on the structure of fluoride salts is closely related to new energy industries.
This work was supported by the National Natural Science Foundation of China and the Young Potential Program of Shanghai Institute of Applied Physics, Chinese Academy of Sciences.
Link: https://pubs.rsc.org/en/content/articlelanding/2023/CP/D3CP01096A