Significant progress in Separation and Recovery of Thorium-Containing Molten Salts via Low-Pressure Distillation
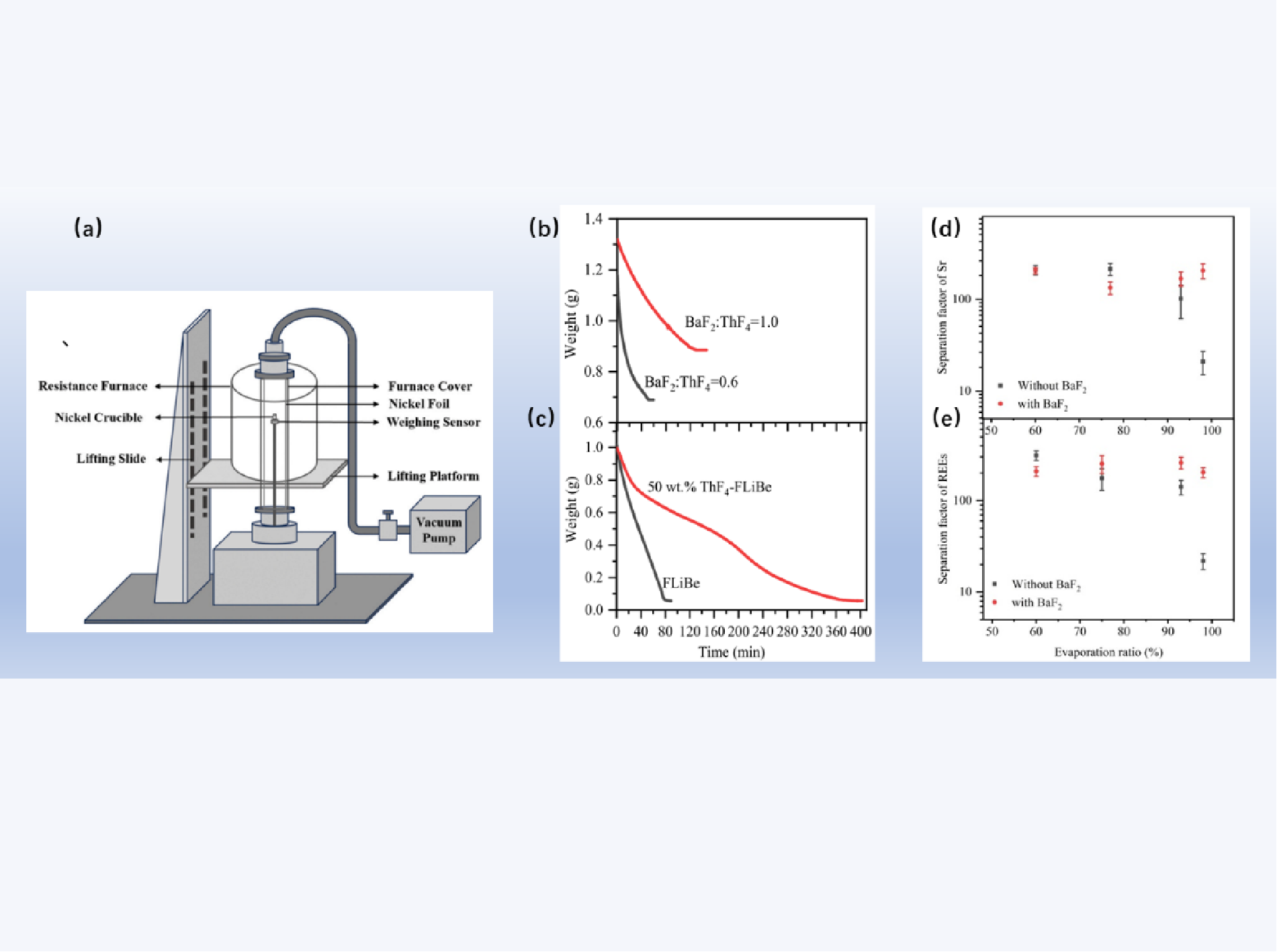
Recently, the researchers at Shanghai Institute of Applied Physics (SINAP), Chinese Academy of Sciences, have made significant progress in Separation and Recovery of Thorium-Containing Molten Salts via Low-Pressure Distillation. The research result, entitled “Separation and recovery of molten salts containing 50 wt.% ThF4 via low-pressure distillation coupled with coordination reaction ” has been published in Separation and Purification Technology (2026, 386: 136507). It was proved that via BaF2-mediated competitive coordination, Li+ in Li3ThF7 complexes is substituted by Ba2+, achieving the release of LiF. The BaF2-introduced competitive coordination not only significantly shortens the separation time of FLiBe molten salt but also enhances the purification efficiency of ThF4 and fission products during the distillation treatment.
In the pyroprocessing of the spent fuel from a thorium-based molten salt reactor (TMSR), the formation of Li3ThF7 complex poses an obstacle to separate and recover LiF-BeF2 (FLiBe) from ThF4-FLiBe mixture salts via the low-pressure distillation. This work aims to facilitate FLiBe recovery by substituting Li+ in the Li3ThF7 complex with Ba2+ through a competitive coordination reaction. The effect of BaF2 addition on the distillation of 50 wt.% ThF4-FLiBe mixed molten salts has been studied. It was found that introducing an optimal amount of BaF2 enhanced FLiBe evaporation speed by over eightfold in 50 wt.% ThF4-FLiBe melts. Correspondingly, the time required for equivalent FLiBe evaporation shortened by nearly 90%, approaching the evaporation speed of ThF4-free FLiBe. At 98% FLiBe evaporation, separation factors for rare-earth and alkaline-earth elements reached 200, nearly tenfold higher than those in BaF2-free systems. By means of ICP-OES and XRD analyses, the real-time evolution of the various elements of Li, Be, Th, and Ba speciation have been revealed during the distillation process. The findings demonstrated that the formation of a stable complex BaThF6 after addition of BaF2 was responsible for the separation and recovery of the mixed salt of 50 wt% ThF4-FLiBe with high efficiency. Molecular dynamics (MD) and thermodynamic calculations indicate that the molten fluoride salt mixture under the conditions of 950 °C and 10 Pa is in good agreement with experimental observations.
The studies of this work suggested the low-pressure distillation technology coupled with the coordination chemical reaction held the potential to open up a broader application prospects for the separation and purification of fluoride molten-salt systems within the nuclear chemical industry.
Full article link: https://doi.org/10.1016/j.seppur.2025.136507